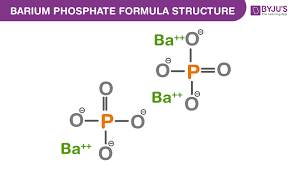
Synthesis of Barium Phosphate
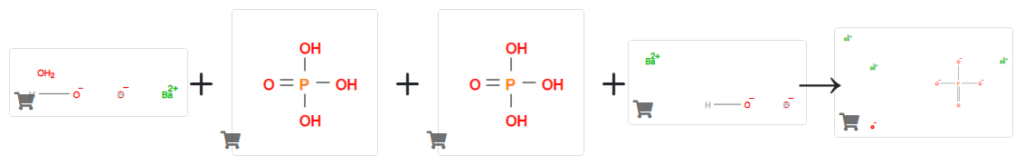
Barium phosphate was prepared by the following procedure. Barium hydroxide (31.23 grams of Ba(OH)2 8H2O, 0.099 moles) was dissolved in 500 grams of deionized water and added to a 1 liter reaction flask fitted with a magnetic stirring bar, reflux condensor and dropping funnel.
The hazy solution was heated to 68° C., and treated dropwise over a 30 minute period with a solution of 7.61 grams of 85%w phosphoric acid (0.066 moles of acid) in 50 grams of deionized water.
The resulting milky white mixture was heated further at 80° C. for an additional two hours, then cooled to 25° C. and filtered using a medium porosity glass filter. The white filter cake was washed several times with deionized water until the wash was approximately neutral pH. The solid was dried under vacuum (80mm Hg) at 80° C. for 24 hours.
Analysis of the product showed 63 %w Ba and 8.9 %w P (theoretical for Ba3 (PO4)2 is 68.4% Ba and 10.3 %w P), with the remainder presumably water.
Literature source US04967016